Learned this in AP Chemistry
What is it?
Intramolecular Forces - attractive forces that hold particles together in ionic, covalent and metallic bonds
- Intra - within
Intermolecular Force - attraction between adjacent molecules
- Much weaker than intramolecular forces
- Forces that hold solids and liquids together
- When a substance undergoes a phase change, intermolecular
Graphs
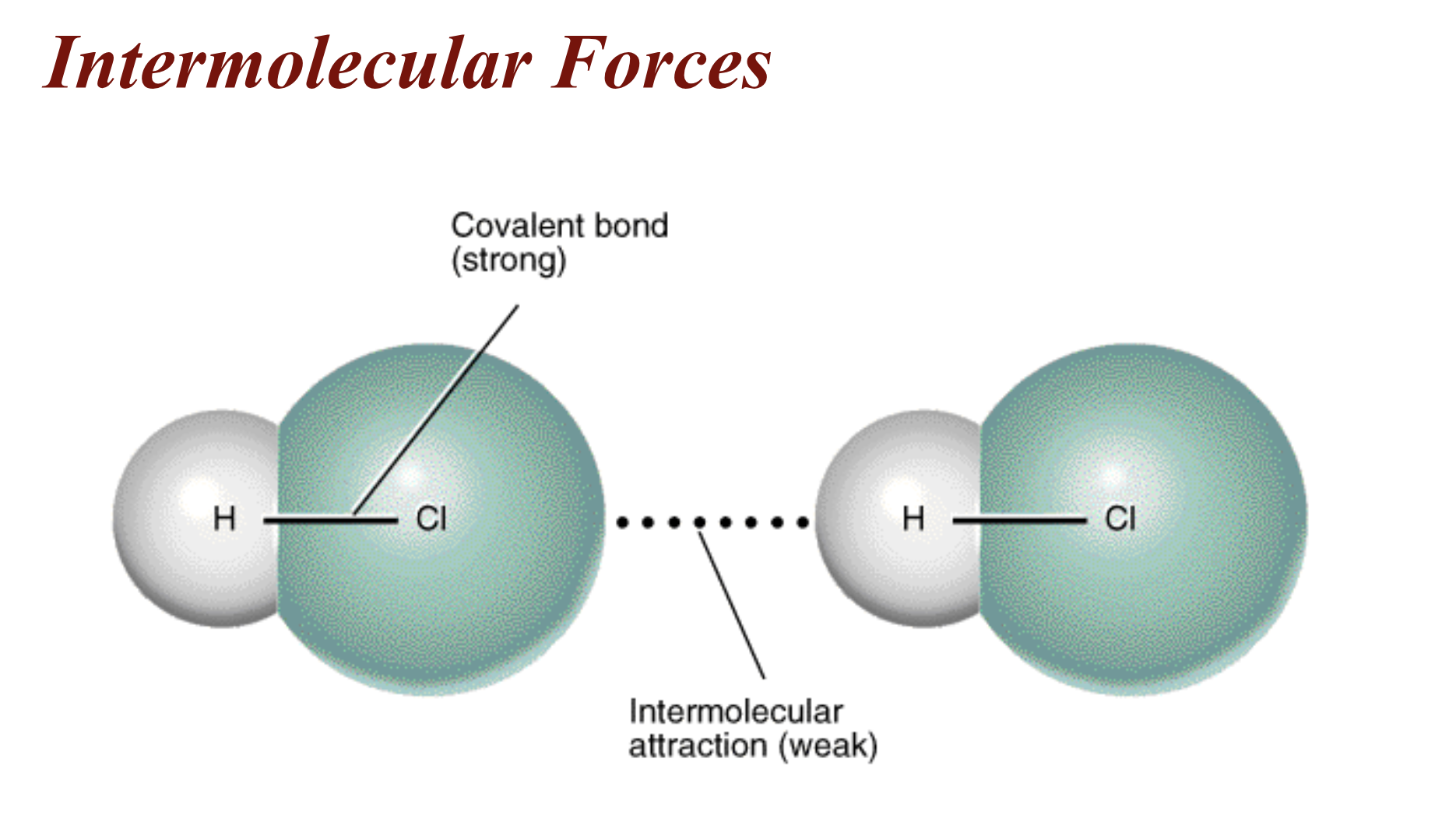
bond energy = 431 kJ/mol intermolecular attraction = 146 kJ/mol
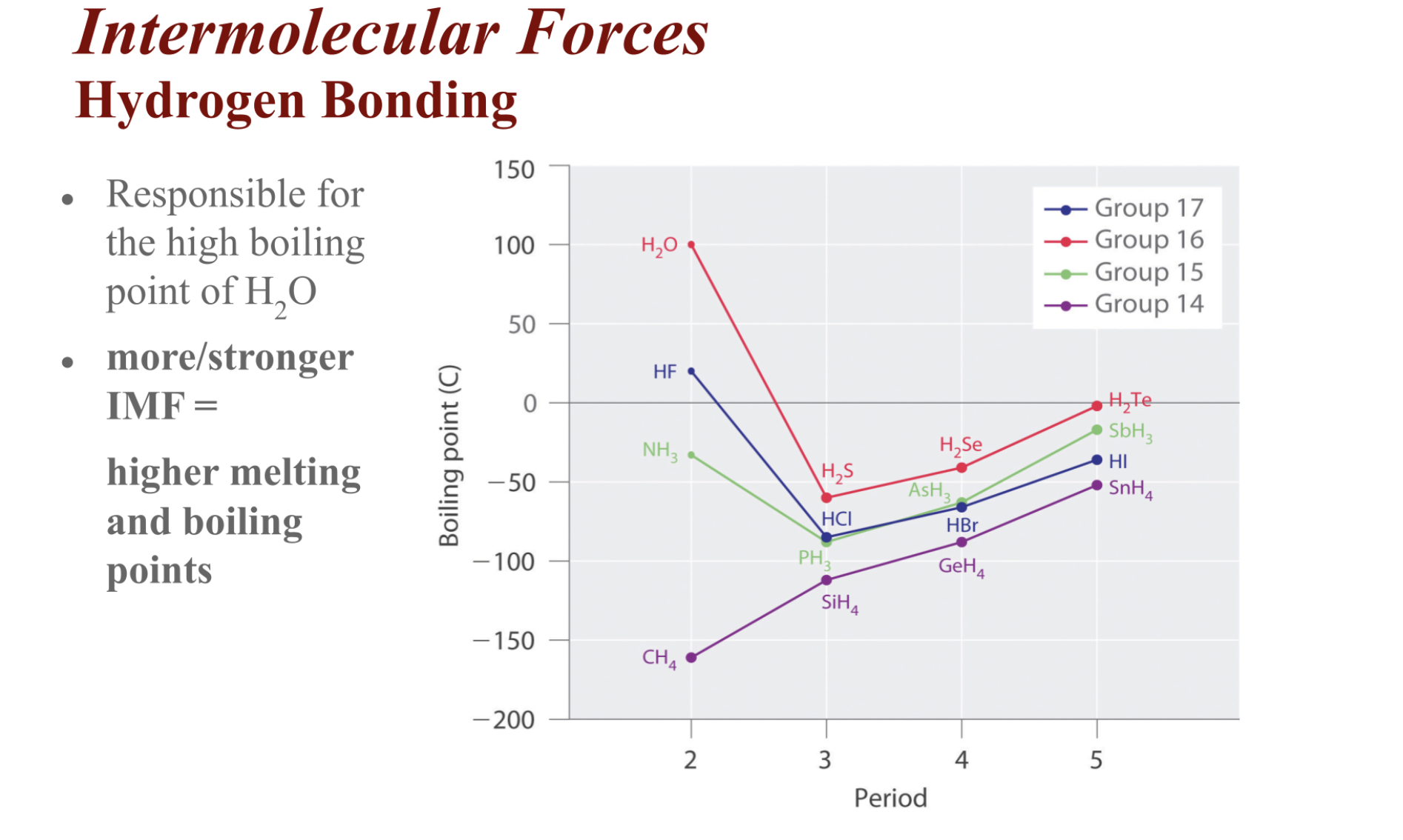
Dipole-Dipole Forces
- Forces that exist between neutral (no charge) polar molecules
- Attraction between
and ends of different molecules
Hydrogen Bonding
- Special case of dipole-dipole forces
- Requires H to be covalently bonded to one of the three most electronegative elements- F, O, or N (hydrogen bonding is FON)
- creates a very polar bond (greater partial
and charges) - Strength:
- hydrogen bonds > dipole-dipole bonds
London Dispersion Forces (LDF)
- weak forces that result from temporary shifts in the density of electrons in the electron cloud
- At given points the electrons are not even distributed
- Results in an instantaneous asymmetric e
distribution - Creates an instantaneous dipole moment
- Results in an instantaneous asymmetric e
- The forces between instantaneous dipoles are called London Dispersion forces
- non-polar is always LDF
- Strengths (strongest to weakest):
- Ionic
- Hydrogen Bond
- dipole-dipole
- LDF
- Polarizability - the ease with which an electron cloud can be deformed
- More electrons = more polarizable -> more instantaneous dipoles -> more LDF’s
- larger molecules (the greater the # of e
) are more polarizable
Trends
The stronger the forces between the particles:
-
the higher the melting point.
-
the higher the boiling point.
-
the lower the vapor pressure.
-
the greater the viscosity.
-
the greater the surface tension.