Introduced to this in AP Chemistry
Rate Laws
- Study the reaction directly after the reaction starts - so you only study reactants
- 2 types of rate laws:
Differential- rate depends on [concentration]- [] references to the molarity of
Integrated- concentration depends on time- Use whichever is easier to experimentally determine
The Differential Rate Law
If the question asks “Rate Law” then it’s referring to Differential Rate Law. Do NOT use Integrated
The rate law expresses the relationship of the rate of a reaction to the rate constant and the concentrations of the reactants raised to some powers
Rate law (DIFFERENTIAL REMEMBER!) can ONLY be determined experimentally
aA + bB -> cC + dD
Rate = k[A]
k = rate constant x and y = calculated values
reaction is x
reaction is y
reaction is (x+y)
Example
F
Rate = k[F
Calculate value of x and y
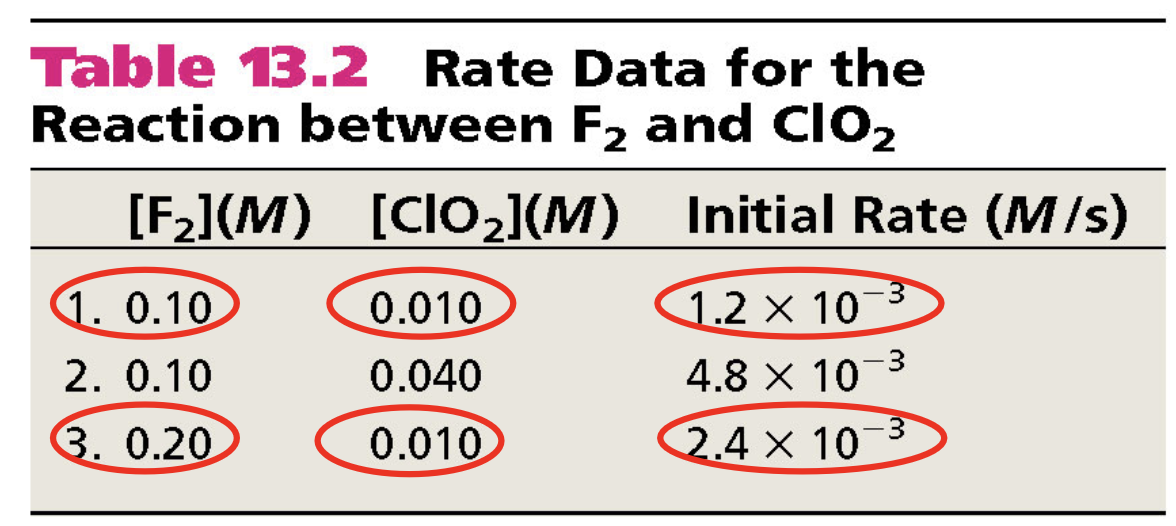
Comparing trial 1 and 3
Double [F
Comparing trial 1 and 2
Quadruple [ClO
Summary
- Reaction order is always defined in terms of reactant (not product) concentrations
- Rate laws are always determined experimentally
- The order of a reactant is not related to the stoichiometric coefficient of the reactant in the balanced chemical equation
Integrated Rate Law
Concentration and time data
Must graph data to determine the order of the rxn
| Order | Linear Graph: |
|---|---|
| Zero | [concentration] vs. time |
| First | ln [concentration] vs. time |
| Second | 1/[concentration] vs. time |
First-Order Reactions
rate = k[A]
After integration you get [A] = [A]
ln[A] = -kt + ln[A]
[A] is the concentration of A at any time t
[A]
The half-life, t
t
Half life does NOT depend on the concentration ([A])
Second-Order Reactions
rate = k[A]
Zero-Order Reactions
rate = k[A]
[A] = -kt + [A]
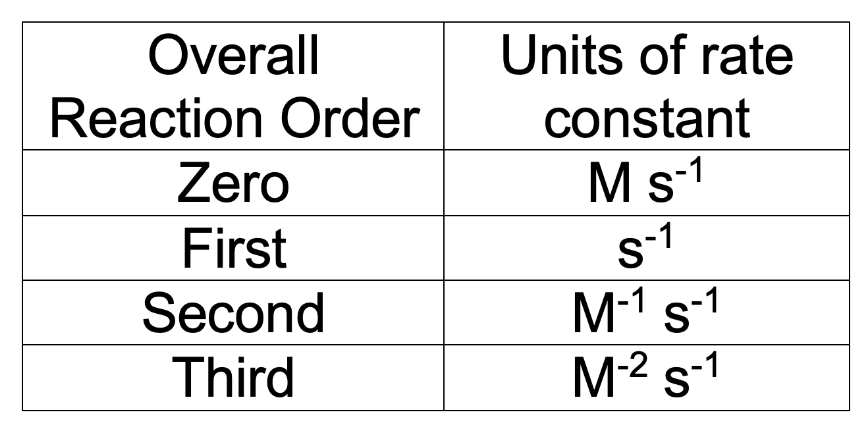
Units for the rate constant
Chemical concept of a reaction
- 0 order = the change in concentration of that reactant does not change the rate of reaction
- 1st order = doubling the concentration causes the rate to double
- n
order = doubling the concentration causes a 2 increase in rate - The rate constant does not depend on concentration