Introduced to this in AP Chemistry
Rules
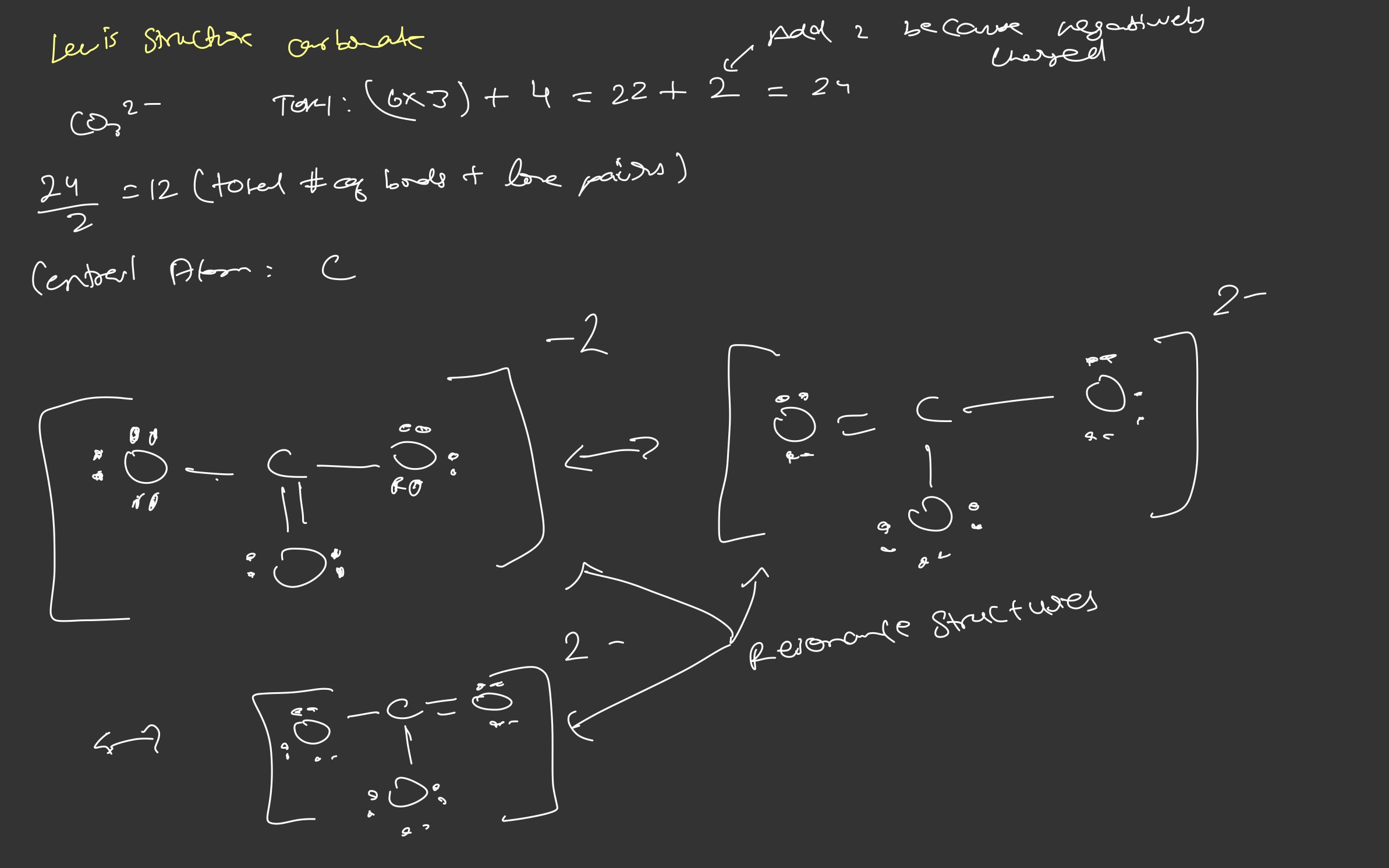
- Add total # of valence e
Add 1 for each negative charge. Subtract 1 for each positive charge - Divide by 2 = total # of bonds + lone pairs
- Determine central atom, place 1 bond between central atom and terminal atom (outside atoms)
- Determine the # of lone pairs for the terminal atoms and place them on atoms
- Complete an octet for all atoms except hydrogen
Localized - This means that electrons belong to covalent bonds between two specific atoms
Resonance structures - structures which can be drawn in more than one way and are separated by double arrows
Resonance structures are a way to represent delocalized electrons that cannot be represented by a single lewis structure
Bond Order
What is it?
Bond order is a measure of the number of shared electron pairs between two atoms in a molecule. It provides insight into the strength and stability of a bond.
Ayush’s terms: Bond order is the number of bonds that are “shared” in the double bond / number of atoms
Example