Introduced to this in AP Chemistry
Useful for determining solubility of ionic solids in an aqueous solution
BaF
K
Solubility vs. Solubility Product
- K
= solubility product - has only one value for a specific temperature - Solubility = equilibrium position concentration: [] = molarity
- K
and molar solubility are not the same thing
Solving Solubility Problems
The salt, BaF
BaF

Solving Solubility with a Common Ion
For the salt Ag
What is the solubility in 0.050 M Na
| Ag | 2Ag | CrO |
|---|---|---|
| Initial | 0 | 0.050 |
| Change | +2s | +s |
| Equilibrium | 2s | 0.050+s |
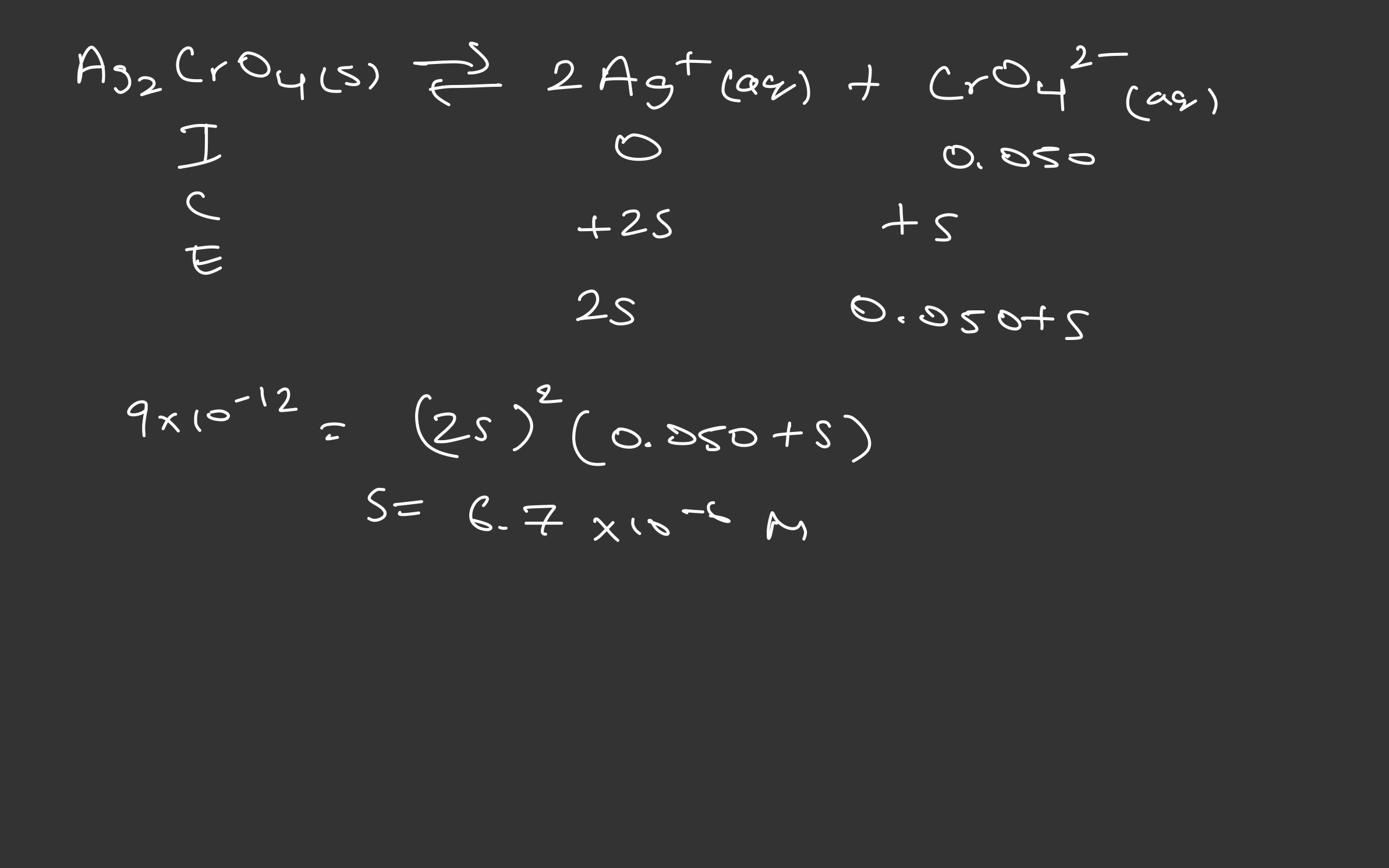 |
Predicting Formation of Precipitate
Q
- K
= Q saturated solution, but no percipitation - K
< Q saturated solution, with percipitation - K
> Q unsaturated solution
Example
If the solutions are added together, predict if a precipitate of PbCl
30.0 mL of 0.10 M NaCl
20.0 mL of 0.025 M Pb(NO
[Pb
[Cl
Q
K
K