Introduced to this in AP Chemistry
What is it?
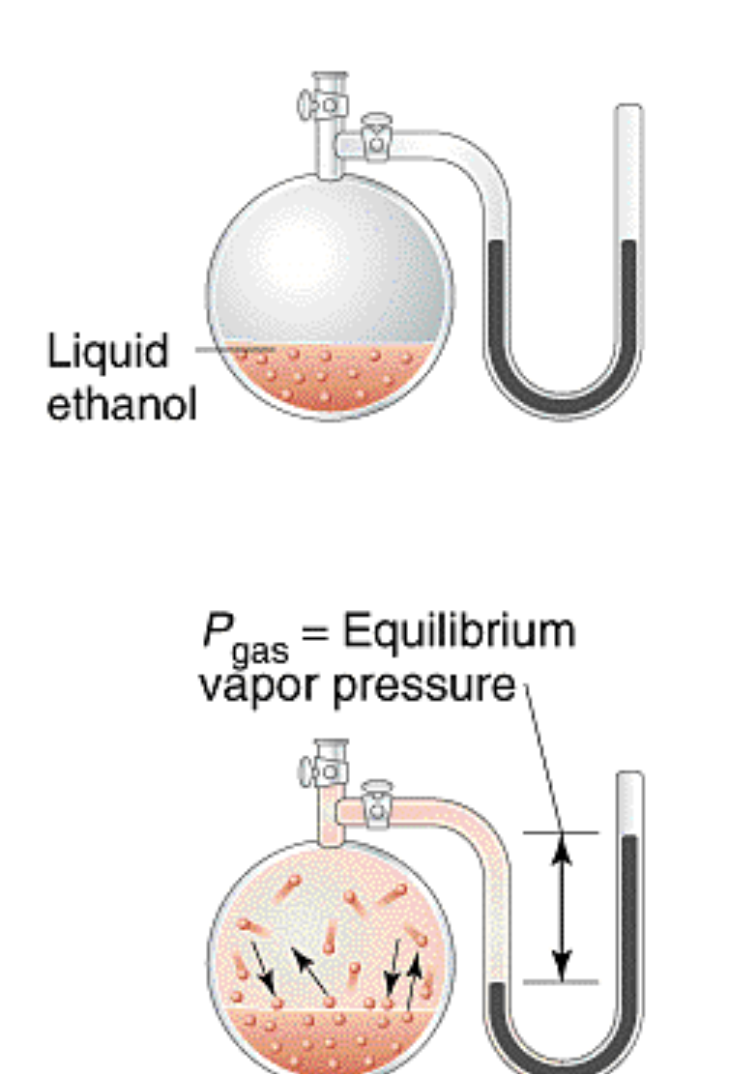
Vapor Pressure: Pressure exerted by the vapor of a liquid (or solid) when condensation and vaporization are in dynamic equilibrium
Vapor pressure is a physical property that’s a result of IMFs
for AP Chemistry we’re going to be focusing on liquids not solids
Rate of vaporization = rate of condensation
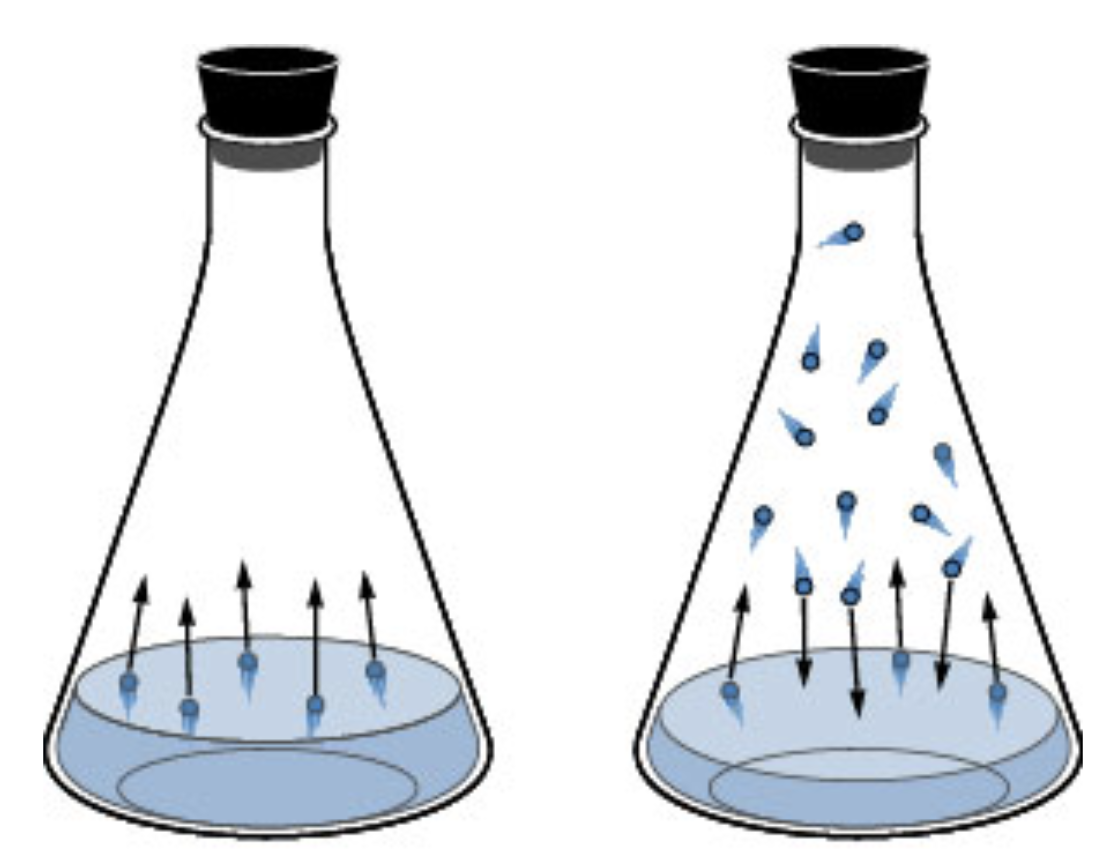
Vapor Pressure (Molecular Level)
Vapor Pressure on the Molecular Level
- Some molecules on the surface of a liquid escape the attraction of the IMFs: liquid -> gas phase
- These gas molecules collide with the container creating a pressure
- After some time the pressure will be constant = vapor pressure
- Dynamic Equilibrium: The point when as many molecules escape the surface as strike the surface and return to the liquid layer
- P
= Height (mmHg)
Volatility and Vapor Pressure
- Volatile substances evaporate rapidly
- Liquid with high vapor pressure
- In general molecules with greater molar mass have lower vapor pressure - why?
- Molecules with higher molar mass need more energy to be evaporated which results in less vapor pressure
- Less vapor pressure means that the IMFs are strong
- Moris’s comments:
- State a specific IMF not a general IMF
- Explain why more molar mass leads to more IMF
- Tie it back to Vapor Pressure
- Sample Answer:
- More polarizable (don’t have to use this) -> more instantaneous dipole moments -> increased LDF -> molecules stay in the liquid phase, so lower vapor pressure